Mibhedha yekurapa inogonawo kunzi mibhedha yekurapa, mibhedha yekurapa, mibhedha yekuyamwisa, etc. Iwo mabhedha anoshandiswa nevarwere kana vari muchipatara. Iwo anonyanya kushandiswa muzvipatara zvikuru, nzvimbo dzehutano dzemataundishipi, nzvimbo dzehutano dzenharaunda, nezvimwe.
Iyo US FDA inoda kuti kana chikafu uye zvigadzirwa zvekurapa zvapinda mumusika weUS, zvinofanirwa kunyoreswa paUS FDA yepamutemo webhusaiti vasati vapinda mumusika weUS.
Mibhedha yemuchipatara inoiswa seKirasi I michina yekurapa muFDA. Iyo US Food and Drug Administration inotsanangura Class I zvishandiso se "zvisina kuitirwa kushandiswa kuchengetedza hupenyu kana kuchengetedza hupenyu, kana kuve yakakosha mukudzivirira kukuvadzwa kuhutano hwevanhu, uye inogona kusapa inogona "Midziyo inoisa njodzi isina musoro yehurwere kana kukuvara.” Midziyo iyi ndiyo inonyanya kuzivikanwa chikamu chemidziyo inodzorwa neFDA, inoverengera 47% yemidziyo yakatenderwa pamusika. Zvishandiso zveKirasi I zvine kubatwa nemurwere kushoma uye zvine zvazvinoita pahutano hwemurwere hwese. Kazhinji, zvishandiso zveKirasi I hazvisangani nenhengo dzemukati dzemurwere, yepakati tsinga system, kana yemwoyo. Midziyo iyi inoenderana nediki remitemo zvinodiwa.
Chitupa cheFDA chezvishandiso zvekurapa chinosanganisira: kunyoreswa kwemugadziri neFDA, kunyoreswa kwechigadzirwa FDA, kunyoreswa kwechigadzirwa (510 fomu rekunyoresa), runyorwa rwechigadzirwa (PMA wongororo), kunyora uye shanduko yehunyanzvi, kubvumidzwa kwetsika, kunyoresa, uye pre-market kuzivisa nezvekurapa uye. midziyo yekuchengetedza hutano, Zvinhu zvinotevera zvinofanirwa kuendeswa:
(1) Makopi mashanu ezvigadzirwa zvakapedzwa zvizere
(2) Dhizaini yedhizaini uye tsananguro yemavara
(3) Kuita uye kushanda musimboti wechigadzirwa
(4) Kuratidzira kwekuchengetedza kana bvunzo zvinhu zvemudziyo
(5) Nhanganyaya yekugadzira maitiro
(6) Pfupiso yemakiriniki ekuedzwa
(7) Mirayiridzo yezvigadzirwa. Kana mudziyo uine radioactive zvinhu kana kuburitsa radioactive zvinhu, zvinofanirwa kutsanangurwa zvakadzama.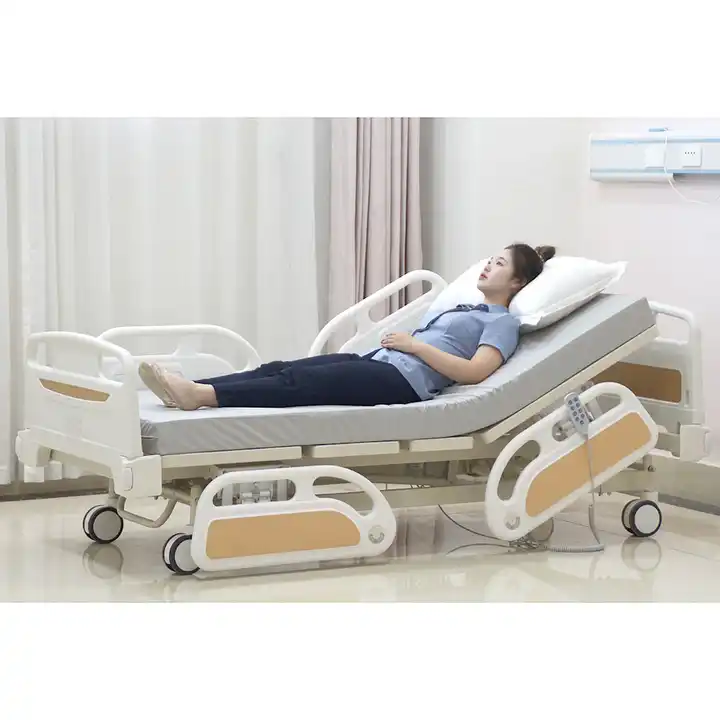
Project Cycle
Nguva kubva pakuongorora kweFDA kusvika pakubvumidzwa kwekupedzisira kazhinji yakareba uye inodzorwa neFDA; kazhinji iyo yese yakajairika process cycle inenge 12 mwedzi
Iyo 510K yekunyorera mibhedha yemuchipatara ndeyotevera:
1. FDA 510(K) zvinyorwa zvekuteerera zvinodiwa
2. Ongororo yakajairika inoshanda kuUS FDA 510k kunyoresa
3. Kusimbiswa kwekuwanikwa kwemagwaro aripo
4. Kuunganidza uye kuenzaniswa kwezvinhu zvakanyoreswa pamusika
5. Gadzirira ruzivo rwechigadzirwa maererano neUS FDA 510k zvinodiwa
6. Gadzirira magwaro ekunyora 510k maererano nemitemo
7. Ita zvidzokororo zvichienderana nemigumisiro yekuongorora yemagwaro ekunyora
8. Zadzisa kunyoreswa kwekambani uye kunyoreswa kwechigadzirwa
taishaninc ine global export certification
Iyo ine 5-yakazara-varidzi vezvikamu
Kuvhara zvinhu zvekuvaka, makemikari, uye maindasitiri emidziyo yekurapa
Isu tiri fekitori ine pasi rose certification, ine mutengo wepagore wekubuda we5,000,000 uye inotumira kune dzinopfuura 160 nyika dzepasirese. Isu tiri yakakura yakasanganiswa maindasitiri epaki fekitori munzvimbo yenzvimbo. Kana zvichidikanwa, ndapota taura nesu munguva uye tumira yakadzama chigadzirwa ruzivo.
Nguva yekutumira: Nov-21-2023




